  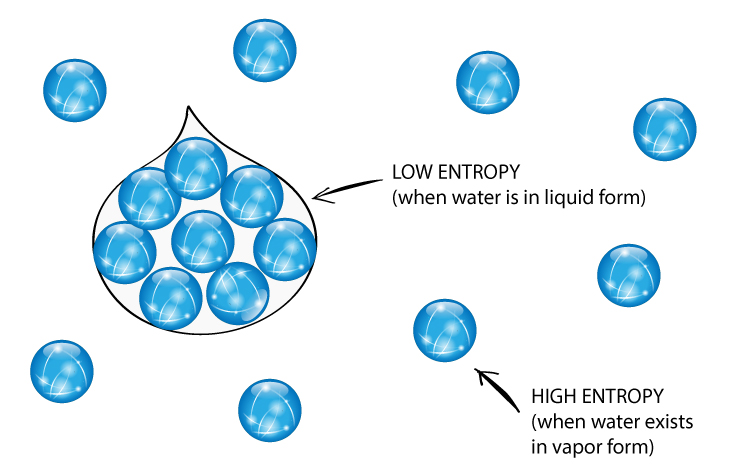
In nonstationary states, the entropy defect similarly acts as a negative feedback, or reduction of the increase of entropy, preventing its unbounded growth toward infinity. Entropy is one of the important concepts in physics and thermodynamics and it is one of the quantities surrounded by amount of mystery. In stationary states, the consequent thermodynamics generalizes the classical framework, which was based on the Boltzmann–Gibbs entropy and Maxwell–Boltzmann canonical distribution of particle velocities, into the respective entropy and canonical distribution associated with kappa distributions. We show that these properties provide a solid foundation for the entropy defect and for generalizing thermodynamics to describe systems residing out of the classical thermal equilibrium, both in stationary and nonstationary states. The entropy defect determines how the entropy of the system compares to its constituent’s entropies and stands on three fundamental properties: each constituent’s entropy must be (i) separable, (ii) symmetric, and (iii) bounded. This defect is closely analogous to the mass defect that arises when nuclear particle systems are assembled. The entropy defect quantifies the change in entropy caused by the order induced in a system through the additional correlations among its constituents when two or more subsystems are assembled. Entropy always increases in irreversible. Entropy stays constant in reversible processes. Entropy is the Greek word for \transformation' Hans C. This paper describes the physical foundations of the newly discovered “entropy defect” as a basic concept of thermodynamics. Entropy is rst de ned by German physicist Clasius, \On various forms of the laws of thermodynamics that are convenient for applications', (1865). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
